
In contrast, Thomson's theory did not provide a clear distinction between the mass distribution within the atom. Rutherford's theory emphasized that the majority of an atom's mass is concentrated in the nucleus. 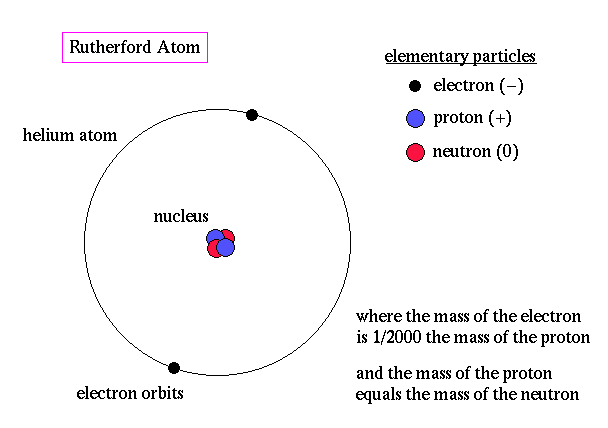
Rutherford's model provided a more accurate representation of the atom's structure, as it explained the deflection of alpha particles and the existence of a dense nucleus. In contrast, Thomson's theory suggested a more uniform distribution of positive and negative charges throughout the atom. Rutherford's theory proposed a concentrated positive charge in the nucleus, with electrons orbiting around it. While both Rutherford and Thomson made important contributions to atomic theory, their models differed in several key aspects: 1. However, it failed to account for the observed deflection of alpha particles in Rutherford's gold foil experiment, which led to the development of the Rutherford atomic theory. Thomson's model was a significant advancement at the time, as it provided an explanation for the existence of negatively charged particles within atoms. Electrons are scattered throughout the atom.Positive and negative charges are evenly distributed throughout the atom.Atoms are composed of a positively charged "pudding" with negatively charged electrons embedded within it.Thomson's theory suggested the following attributes: It was based on his experiments with cathode rays, which led to the discovery of electrons. Thomson's atomic theory, also known as the plum pudding model, was proposed in 1904. It provided a more accurate representation of the atom compared to Thomson's model. Rutherford's theory revolutionized the understanding of atomic structure by introducing the concept of a concentrated positive charge in the nucleus and the idea that electrons orbit the nucleus. Electrons are negatively charged particles. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
